 So a higher shift as you move to the left. So as you go to the right, you're talking about a And the chemical shift would be, would be a similar idea to the frequency. And so instead of talking about frequency, we could talk aboutĬhemical shift values here. TMS are more shielded than almost all organic compounds. TMS is actually our standard because the protons on And so therefore, the protons on benzene have a higher frequency signal than the protons on TMS. Left on an NMR spectrum, you're talking about a So a lower frequency signalĪs you move to the right on an NMR spectrum. In an earlier video we said that as you go to the right on an NMR spectrum you're talking about a And here's the signal for TMS right here. And the protons on TMS areĪll in the same environment and so therefore we wouldĮxpect one signal for TMS. 
If we compare benzene to this compound, this is tetramethylsilane or TMS. And so here's the one signal on a spectrum due to the protons on benzene. Were chemical equivalents, and should give us only one Were in the same environment, therefore all six protons Use a window function such as a small negative line broadened exponential function, or a gaussian function, and you can often split these lines almost to the baseline.- In the previous video, we looked at the protons on benzene. You can get a better understanding of these small splittings if you use resolution enhancement when you process your data. These are often not reported in experimental data, because it is quite difficult (or at least time consuming) to resolve all of these couplings. So what you are seeing is genuine splitting coming from small couplings. (And we'll leave alone the discussion about whether they have reported apparent splittings, as opposed to J couplings, otherwise J56=J65), Again, we see this, and each of these of these should be split due to the small meta couplings. But, we also expect to see some meta coupling to H3 (again 1-3Hz). That gives us the reported triplet, which is in fact a dd. For the H5, we expect, again, a large ortho doublet coupling to H6, but also a large ortho doublet coupling to H4 (again, ~7-8Hz). On a good shimming day, in better solvents than dmso, we might even expect to see para splitting (<1Hz - don't hold your breath though). So, take a deep breath in.įor Salicylamide, as described in your paper, we would expect H6 to be a large doublet (ortho to H5, ~7-8Hz), but also show meta coupling to H4 (~1-3Hz), and so we expect this large doublet to exhibit small splittings. This is where an understanding of the expected coupling pattern for the molecule is very helpful. Splitting due to coupling Often it is difficult to fully resolve small couplings, especially those that come from aromatic systems or long range pathways, where the size of the splitting might be about 1Hz or so.This could account for this type of spectrum (but more than likely doesn't), and that is why you would do other experiments such as COSY or HSQC to verify this. Obviously, the relative populations will determine how they actually appear. If two singlet species have a non-identical chemical shift within their natural linewidths, then they will appear as a shouldered peak. NMR spectra are simply the superposition of all nuclei in the sample.  
If this lineshape was genuinely due to a shimming anomaly, that indicates a pretty serious z shim issue, and you've probably got some floating solid in the sample. As an aside on this spectrum, these 'shoulders' you are looking at aren't really shoulders - they are split peaks. Always check these small molecule peaks when you run your spectra to determine shim quality. 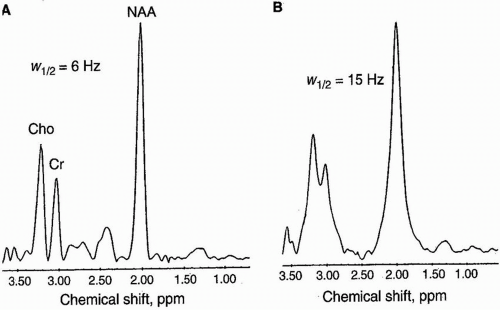
They will all have exactly that same lineshape anomaly, with identical shoulders. So, have a look at the TMS peak, or solvent peak, or that internal reference of ethyl acetate that your supervisor suggested was an impurity. Fairly common, but the key to diagnosing this cause is that it will affect all peaks in the same way (within the natural linewidth of the peak). Shoulders can come about from typically three origins: When a peak tends to deviate from the standard Lorentzian, we might describe this as having a shoulder. The term 'shoulder' is really an informal description used to describe the appearance of parts of peaks in the NMR spectrum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
